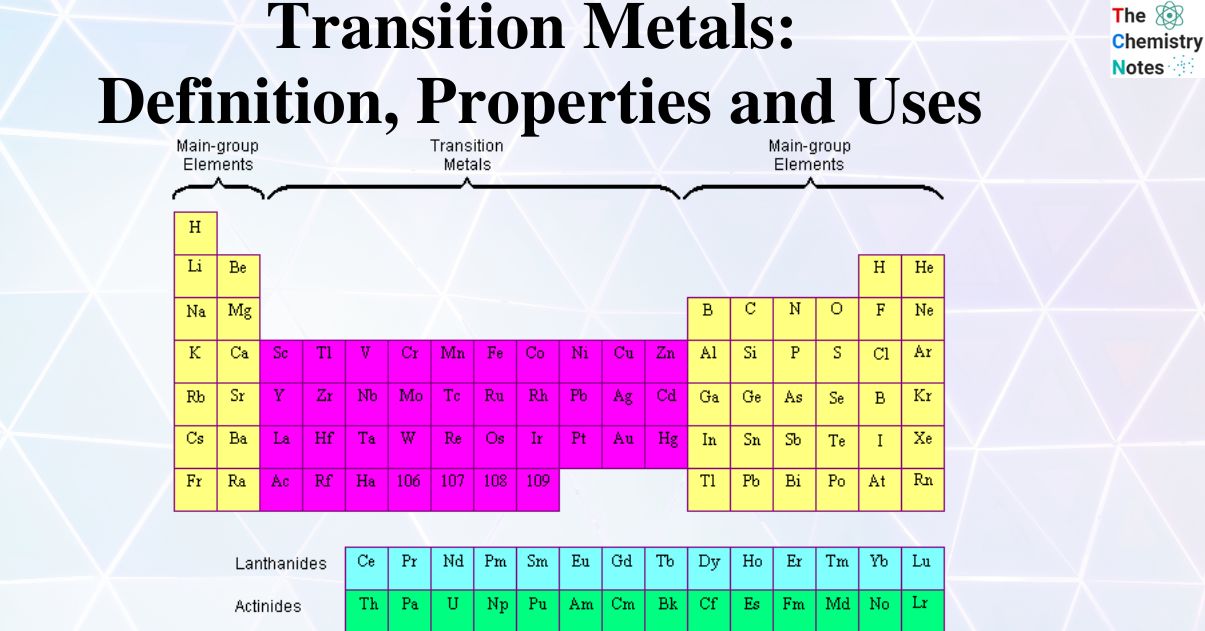
List of colors of transition metal ions and ion complexes Ĭertain transition metals like iron, copper, titanium, and mercury can be found in the earth’s crust, with iron being the most abundant. Color is perceived as the radiation transmitted by these compounds. When white radiation falls in these complex compounds, the d electron absorbs some radiation, and the rest is transmitted. In complexes of transition metals, these d orbitals have different energies. In the d-d transition, an electron moves from one d orbital to another. A charge transfer transition occurs when the metal is in a high or low oxidation state, during which charge is transferred between the metal and a nonmetal species (ligand).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
